
Ring of refined 99.96% pure plutonium, mass 5.3 kg, 11 cm diameter (sufficient to manufacture the fission core of a nuclear weapon). This image, by the U.S. Department of Energy, is in the public domain.
by John Walker
Elsewhere we've explored uranium enrichment, a dual-use technology which can be used either to enrich natural uranium for use in civil nuclear power reactors or, through further enrichment, for use in nuclear weapons. Now, let's examine the other path to the bomb: the production of plutonium from natural uranium by irradiation in a nuclear reactor and chemical separation. Let's begin with some basics.
If you want to build a nuclear fission weapon (either for use by
itself or as the trigger for a thermonuclear
weapon), you need a critical mass of
fissile material which is kept in a sub-critical configuration until
the weapon is to be detonated, then rapidly assembled into a
critical configuration where the nuclear chain reaction can run
away, producing an explosive yield. A variety of elements and isotopes can theoretically be used
in nuclear fission weapons, but since the start of the nuclear age,
only two have ever actually been employed in weapons: uranium-235 (U-235) and
plutonium-239 (Pu-239). Uranium exists in nature, but only 0.711% of
natural uranium is U-235, so in order to build a bomb the costly and
difficult process of
enrichment must be performed to separate the U-235 from the
99.284% of U-238, which cannot sustain the fast chain reaction
required in a weapon.
Plutonium does not exist in nature. The half-life of Pu-239 is just 24,000 years, so even if any were present when the Earth formed, it would have long ago decayed to other elements. But if you take U-238, which makes up the overwhelming fraction of natural uranium and bombard it with neutrons, some nuclei will absorb a neutron, transforming them into U-239 (a nucleus with the same 92 protons as U-238 but an additional neutron). But this nucleus has too many neutrons to be stable, and decays with a half-life of 23.5 minutes by beta decay: the emission of a beta particle (electron), transforming one of the neutrons in the nucleus to a proton, which transmutes the U-239 into element 93, Neptunium, yielding the isotope Np-239. Np-239, while more stable than U-239, remains unstable and with a half-life of 2.36 days, also undergoes beta decay, resulting in Pu-239 which, with its half-life of 24,000 years, can be considered as stable on the human time scale. Here is how nuclear chemists write this sequence of reactions.
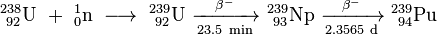
This seems pretty simple. All we have to do is obtain natural uranium, which is mostly U-238, bombard it with neutrons, wait a while, and we'll end up with plutonium which, being a distinct chemical element, can be separated from uranium by a series of chemical reactions we'll discuss below. So where do we get some neutrons? The first atoms of plutonium were made in a cyclotron at the University of California, Berkeley, but this process is so slow and inefficient it could not produce sufficient quantities of plutonium to be visible to the human eye, not to mention the kilograms required to make a bomb.
But a nuclear reactor also produces abundant neutrons, with enough left over from the fissioning of U-235 to irradiate U-238 and start the sequence of reactions which will yield Pu-239. Furthermore, by using a neutron moderator of graphite or heavy water, it is possible to build a nuclear reactor which will sustain fission using natural uranium, eliminating the need for uranium enrichment. (For reasons of cost and efficiency, most nuclear power stations use regular light water as moderator and coolant, but this requires enriched fuel.)
In principle, you build a graphite or heavy water moderated reactor, fuel it with natural uranium refined from uranium ore, start it up, let it run for a while, then remove the fuel elements, which will now contain Pu-239 bred from the U-238 in the natural uranium you started with, and chemically separate the plutonium and hand it off to the bomb builders. In practice there are a large number of messy details which complicate the process. While plutonium for weapons purposes is usually produced in dedicated plutonium production reactors using natural uranium fuel, any nuclear reactor which has U-238 in its core (which includes all civil power reactors) will breed plutonium as it operates. For reasons we'll discuss below, unless the reactor is operated in a very inefficient manner for power production, this plutonium is much less suitable for building bombs, but it can be diverted for weapons use, which requires careful monitoring of spent power reactor fuel by the International Atomic Energy Agency (IAEA).
As U-238 is irradiated by neutrons in a reactor, Pu-239 is produced by the process described above, but that isn't the end of the story. In a neutron-rich environment, Pu-239 can capture an additional neutron and be transformed into the Pu-240 isotope. Pu-240 is chemically identical to Pu-239, but has a shorter half-life of 6,563 years and, more importantly, undergoes spontaneous fission at a rate 41,500 times greater than that of Pu-239. All of these spontaneous fissions release neutrons, which can provoke predetonation (or a fizzle), in which the nuclear weapon blows itself apart before the intended explosion occurs. This neutron background from Pu-240 rules out the simple gun assembly weapon design possible with U-235, and requires a much more complicated and difficult to perfect implosion design. The higher the degree of contamination of the plutonium with Pu-240, the more sophisticated the weapon design must be to avoid a fizzle.
In order to minimise the amount of Pu-240, fuel rods in a plutonium production reactor should be irradiated for a relatively short period: long enough to transmute around 1% of the U-238 into Pu-239, but not so long that too much Pu-239 is converted into Pu-240. Plutonium with less than 7% of Pu-240 is considered “weapons grade”. In a power reactor, fuel elements are left in the reactor much longer, and plutonium extracted from their fuel rods may have 18% or more Pu-240—this is called “reactor grade ” plutonium. This doesn't mean you can't make a bomb from reactor grade plutonium: in 1962, the U.S. conducted a nuclear test of a bomb using plutonium with a high Pu-240 fraction, although to this day the precise isotopic composition of the plutonium has not been disclosed.
Now let's turn to how plutonium is extracted from the fuel irradiated in a reactor. The irradiated fuel rods removed from the reactor are a highly radioactive witches' brew of uranium, plutonium, and fission products, many of the latter intensely radioactive and emitting large amounts of heat. Fortunately, the most radioactive fission products necessarily have the shortest half-lives, so the fuel rods are first dumped into a holding pool filled with water and left there until the most intense radioactivity subsides.
Next, the fuel elements are dissolved, usually in nitric acid, which results in a liquid containing all of the contents of the rods and their cladding. Any material which did not dissolve is filtered out after this step. Then, the fission products are separated from the uranium and plutonium (which have similar, but not identical chemistry) by a chemical process which exploits the fact that uranium and plutonium are soluble in organic solvents, while the fission products are not and remain in the aqueous solution. The fission products are removed, to be stored as high-level nuclear waste, leaving a mix of uranium and plutonium in the organic solvent. This is then treated with aqueous ferrous sulphamate, which separates the plutonium from the uranium, which can then be extracted and sent to subsequent processing steps. This is a gross over-simplification of the PUREX process which is used for industrial-scale nuclear fuel reprocessing, but these are the essentials. The following video demonstrates this process using real spent nuclear fuel at the laboratory scale.
It is one thing to do this in a glove-box and entirely another to undertake it with tonnes of material, especially considering that if you allow a critical mass of plutonium (10 kg or less depending on circumstances) to accumulate anywhere in the process, you'll have a really bad day. So intense is the radiation from spent fuel rods that the process must be performed in heavily shielded hot cells by remote control. Here is a video (with marginal image quality in some parts) describing industrial-scale plutonium production at the U.S. Hanford Site.
Unlike uranium enrichment, which can be done either for nuclear power or weapons use, plutonium production on a large scale really only has weapons applications. A country with a large civil nuclear power programme may undertake fuel reprocessing in order to recover plutonium for use in power reactors as MOX fuel, but there is no need for a nation with a fledgling nuclear power program to reprocess: it is easier and more economical to ship spent fuel to a country with a reprocessing infrastructure such as Britain, France, or Russia, and receive ready-to-use MOX fuel in return, all under IAEA safeguards.
For nuclear weapons, especially those such as ballistic missile warheads where minimising mass is important, plutonium is the way to go. Its critical mass is around 1/5 that of U-235, which scales everything down in volume as well as mass. Designing weapons with plutonium is more difficult, but plutonium can be produced starting from natural uranium in compact facilities which are easier to conceal and protect against enemy action, as opposed to uranium enrichment plants which are extravagantly large and require materials which, when procured on the open market, raise eyebrows.
Plutonium may be easier to get, but it's harder to use. The weapon design is more demanding and, compared to a uranium gun bomb, without testing it's difficult to be sure the bomb will go boom. While it used to be said that every country which had tried to build a nuclear weapon had succeeded on the first try, this is no longer the case: the North Korean 2006 nuclear test, believed to be a plutonium implosion device, appears to have had a fizzle yield of 0.48 kilotons. Basing one's strategy on nuclear deterrence with plutonium weapons which have never been tested is risky in the extreme, although demonstrating the infrastructure may create sufficient ambiguity that adversaries are disinclined to roll the dice.
Here's the summary for policy-oriented readers who made it this far. There are two paths to nuclear weapons: uranium enrichment and plutonium production. Uranium enrichment requires a large industrial infrastructure which is relatively easy to detect by monitoring its supply chain but which can be argued to be intended purely for a civil nuclear power program. Plutonium production can be done with a natural uranium reactor which requires no fuel enrichment (but a supply of reactor-grade graphite or heavy water, which can be monitored) and a chemical plant of modest size. It is technically easier to build weapons from uranium than plutonium. The simplest uranium bomb designs are hideously inefficient, but are essentially guaranteed to work without testing. Plutonium bomb designs are more difficult, and it's much more risky to rely upon them without a test, which will disclose a nation which conducts it as being a nuclear weapons state. International safeguards against nuclear weapons proliferation must monitor both paths to nuclear weapons, and need to look for the very different signatures of the uranium and plutonium paths.
This is a lecture on how IAEA safeguards work, and their limitations in practice.
Acknowledgement: This essay has been informed by E-mail discussions with Robert G. Kennedy, III, who resides in America's Atomic City and knows far more about these matters than this scrivener. Any errors are, of course, my own. Mr Kennedy coined the observation, “Plutonium: easy to get, hard to use. U-235: hard to get, easy to use.” which I quoted in part above.
|
|
This document is in the public domain.